Vol. 2 (2019), Article ID 236074, 5 pages
Research Article
Anti-Cancer Screening of Some Transition Metal Ion Complexes with Coumarin Derivatives
Dalal M. Ibrahim,1 Juliana Jumal,2 and Wedad Melad Al-Adiwish3
1Chemistry Department, Faculty of Science, Omar Al-Mukhtar University, P.O. Box 919, Albyda, Libya
2Faculty of Science and Technology, Universiti Sains Islam Malaysia, Bandar Baru Nilai, 71800 Nilai, Negeri Sembilan, Malaysia
3Chemistry Department, Faculty of Science, Azawia University, P.O. Box 16418, Az-Zawiyah, Libya
Received 9 March 2019; Revised 15 April 2019; Accepted 16 April 2019; Published 8 May 2019
Dalal M. Ibrahim, Juliana Jumal, and Wedad Melad Al-Adiwish, Anti-Cancer Screening of Some Transition Metal Ion Complexes with Coumarin Derivatives, Journal of Transition Metal Complexes, 2 (2019), art236074. doi:10.32371/jtmc/236074
In this study, an equal amount of -4-methyl-5,7-(bis p-chlorophenyl azo) coumarin (ligand 6-hydroxy) and copper, cobalt, and nickel (metals) were reacted to synthesize the respective metal ion complexes. The synthesized metal complexes were characterized using various approaches such as CHN elemental analysis, 1H-NMR, mass spectral data, and FTIR. The outcome of the spectroscopic analysis showed the coordination of the ligand to the metal ion that is in the complexes through hydroxyl group; the data also showed the complexes to exhibit octahedral geometry. Further studies on the solid complexes were performed by molar conductance where all the synthesized metal complexes exhibited nonconductive properties in chloroform. An MTT-based cytotoxicity screening of the complexes against lung and breast cancer cells showed Co(II), Cu(II), and Ni(II) complexes to exhibit cytotoxic activity against the studied cells at low concentrations.
bis-coumarins complexes; anti-cancer screening
The metal ions-binding capacity of coumarins presents an extra means of manipulating
their pharmacological response.
Transition metals complexes have gained much research interest, especially 3d transition
metal ions which are significantly
important in coordination and bioinorganic chemistry. They can form chelates with
several cations; this chelates formation
is indicated by a color change at a given pH. The formation of the chelates is characterized
by the high stability of the
product due to the formation of 6-membered rings. Monohydroxy compounds that contain
coumarin have shown great importance
owing to their increased chelates-formation tendency when azo groups are introduced
to such hydroxyl coumarins; the
presence of these hydroxyl substituents also improves the potency of the coumarin
rings in the compounds [1].
The biological activity of some 3d transition metal ions in biological systems has
been reported; these ions are often
referred to as metaloproteins because they are sites for the activity of most enzymes
and also determine the structure
of most active sites. The partially filled d orbitals of transition metals confer
them the ability to exhibit several
oxidation states. Their mechanism of their cytotoxic activity has been investigated
in a bid to develop new antitumor
agents. Some coumarin metal complexes have in some cases shown higher biological activities
compared to their ligands
[2,3,4,5].
Nowadays, several studies have reported the biological activities of coumarin complexes with metals. Metal ions' binding to coumarins has been reported to enhance the biological activity of such complexes. To understand the factors responsible for the biological activity of coumarin derivatives, it is necessary to investigate their binding properties to different metal ions [6,7,8,9,10]. Here, we report new experimental studies on the biological activity of complexes with coumarin derivatives.
All reagents used to synthesize the complexes were commercially sourced and of the
highest level of purity.
A solution of p-chloro aniline was prepared and cooled in ice prior to diazotization
with 20 mL of 0.01 M
aqueous solution of sodium nitrite. Then, the diazonium solution (0–5 ℃) was gradually
introduced into 0.01 M 6-hydroxy-4-methylcoumarin solution prepared in a solution containing ethanol and sodium hydroxide (100 mL). A schematic representation of the reaction process is presented in Figure 1.
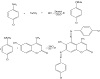
Figure 1: The reactions during the formation of 6-hydroxy-4-methyl-5,7-(bis p-chlorophenyl azo) coumarin.
The process of the solid chelates synthesis involved the mixing of a hot alcoholic
0.001 M saturated metal ion solution with the pre-determined amount of each ligand which is enough to form 1:1
molar ratio complexes. A dilute ammonia solution (1:10) was used to maintain the pH of the solution at a range of
6.5–7.5 [11]. The resulting
mixture was heated with occasional stirring on a steam bath for 4 h and later to dryness.
The complex resulting from the solution was then dissolved in ethanol, filtered via suction, and washed
again with ethanol to get rid of the unreacted materials. The final colorless filtrate was obtained preserved in a
vacuum desiccator for further use.
The schematic illustration of the chemical reaction during the formation of coumarin complexes is shown in Figure 2.
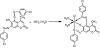
Figure 2: Schematic illustration of the reactions during the formation of coumarin complexes.
Most of the in vitro assays of the response of cells to external factors are based
on the measurement of the viability
and proliferation of such cells when exposed to such factors. One of the widely accepted
methods of cell proliferation
studies is the tetrazolium salts reduction method which involves the reduction of
the yellow tetrazolium MTT by
dehydrogenase enzymes to NADPH and NADH. The formazan formed from the dehydrogenase
activity is then solubilized
and spectrophotometrically quantified. This method of cell proliferation assay measures
the rate of cell proliferation
and the metabolic events that can result in cell death (apoptosis) or necrosis. To
hasten sample handling, the number
of assay steps during MTT-based cell proliferation assay has been minimized as much
as possible. During MTT-based assays,
the MTT reagent contributes less to the absorbance values, and for each cell type,
there is a linear relationship between the number of viable cells and the color produced. This allows an accurate determination
of the rate of changes in cell proliferation.
The cytotoxicity activity of the compounds produced in this study was evaluated using MTT assay against breast cancer (MCF-7) and lung cancer (A549) cells. The cells were plated at a cell density of 2×105/mL in a 96-well culture plate before introducing the complexes at different concentrations of 0.468 μM/L, 0.936 μM/L, 1.875 μM/L, 3.75 μM/L, 7.5 μM/L, 15 μM/L, and 30 μM/L. The cells were allowed to incubate for 72 h at 37 ℃ + 5%CO2; the control cells were left untreated. After 72 h,
the cells were washed twice with PBS before adding 10 μL of 0.5 mg/mL MTT solution into the wells. The cells
were further incubated for another 4 h at 37 ℃ + 5%CO2 before dissolving the formed formazan
crystals in 100 μL of dimethyl sulfoxide (DMSO). The absorbance of the formed purple color was determined
at 570 nm in an ELISA reader.
The synthesized complexes were characterized and found to be stable in air, soluble in CDCl3 and DMSO, but insoluble
in ethanol. The CHN elemental analysis shown in Table 1, the FTIR data shown in Table 2, the 1H-NMR data shown in Table 3, and the mass spectral data showed a successful synthesis
of the investigated complexes. From the molar conductance studies, all the complexes were observed to be neutral and
nonconductive. The FTIR spectra showed a shift in the observed band for the vibration
of the N=N bond in the
complexes compared to the free ligands to a lower wavelength, indicating the site
of the chelation process. The upward shift in the OH band (peaked at 3,339 cm−1 and 3,600 cm−1 in the free
ligands and complexes, resp.) indicates coordination via the hydroxyl group. The observed
band at the wavelength range of 3,600 cm−1 indicates the vibration of the OH group of the water of hydration and water of
coordination. The bands assigned to the carboxyl group are the most important bands in the free ligands group; these
bands shifted to a lower wave number (in the range 1,199 cm−1) due to complexing with C-O.
In the 1H-NMR report, the obtained data showed the absence of OH (which appeared at 12.54 ppm
in the free ligands) in the Ni complex, thereby confirming the deprotonation of OH. It
also confirmed that the ligand
coordinated to the metals via the hydroxyl group.
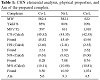
Table 1: CHN elemental analysis, physical properties, and Λm of the prepared complex.

Table 2: Some of the significant IR bands exhibited by the complexes.

Table 3: 1H-NMR spectral data of the investigated Ni complex.

Table 4: IC50 values of ligand and complexes in MCF-7 and A549 cell lines.
In the spectra of the complexes, the pyrone ring exhibited the signals observed at 6.60 ppm while those observed at 7.73–7.36 ppm were assigned to the protons of the aromatic ring as shown in Figure 3.

Figure 3: 1H-NMR spectrum of complex (Ni-L).
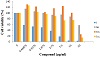
Figure 4: Effect of ligand L and complexes on cell viability of breast cancer cells MCF-7.

Figure 5: Effect of ligand L and complexes on cell viability of lung cancer cells A549.

Figure 6: Effect of ligand L on cell viability of breast cancer cells MCF-7.

Figure 7: Effect of Cu-L complex on cell viability of lung cancer cells A549.

Figure 8: IC50 value of L on MCF-7 cells.

Figure 9: IC50 value of CuL on A549 cells.
At 30 μg/mL, the ligand exhibited cytotoxic activity than the complexes (Cu, Co, and Ni) with values of 2.81%
and 65.44% for MCF-7 and A549, respectively. Some coumarins which contain halogen
groups are highly cytotoxic [12].
Thus, the presence of p-chloro substituents plays important roles in the antibreast
cancer activity. Against the lung
cancer cells A549, ligand L showed weak cytotoxicity; therefore, p-chloro substituent
is selectively active
against MCF-7. Moreover, the cell viability of the three complexes of Cu, Co, and
Ni against MCF-7 was 14.57%, 26.09%,
and 57.54%, respectively. According to the obtained results, the Cu complex was the
most cytotoxic agent against MCF-7
cells compared to the tested Co and Ni complexes.
Cu complexes exhibited a high cytotoxicity against MCF-7 and A549 while the other complexes showed moderate to weak activities against both cell lines. Furthermore, the IC50 results signified that the ligand is the
most active against MCF-7 (at the concentration of 1 μg/mL) while Cu complex is the most active
against A549 (at the concentration of 1.875 μg/mL).
The synthesized coumarin-metal complexes in this study showed the formation of their
structure through the binding of
the nitrogen atoms of the bis azo group in the ligands with the OH group of the aromatic
ring in the coumarins. The
synthesized complexes were characterized by 1H-NMR. The complex formation resulted in the disappearance
of the OH group in the free ligands. The antiproliferative activity of the synthesized
coumarin-metal ion complexes was determined and the results showed a significant activity of the complexes against
the studied cell lines.
The authors declare that they have no conflict of interest.
- I. Kostova, Synthetic and natural coumarins as cytotoxic agents, Curr Med Chem Anticancer Agents, 5 (2005), 29‒46.
- E. G. H. Shahinian, I. Haiduc, and I. Sebe, Synthesis and characterization of new azo coumarin dyes, UPB Sci Bull B, 73 (2011), 154‒160.
- A. L. El-Ansary, E. M. Ebeid, and M. M. Omar, Emission characteristics
of some 8-(arylazo)-7-hydroxy-4-methylcoumarins, Spectrochim Acta A Mol Spectrosc, 43 (1987), 709‒710.
- O. M. Abdelhafez, K. M. Amin, R. Z. Batran, T. J. Maher, S. A. Nada, and S. Sethumadhavan, Synthesis, anticoagulant and PIVKA-II induced by new 4-hydroxycoumarin derivatives, Bioorg Med Chem, 18 (2010), 3371‒337.
- I. Kostova, Coumarins as inhibitors of HIV reverse transcriptase, Curr HIV Res, 4 (2006), 347‒363.
- I. Kostova, G. Momekov, M. Zaharieva, and M. Karaivanova, Cytotoxic activity of new lanthanum (III) complexes of biscoumarins, Eur J Med Chem, 40 (2005), 542‒551.
- I. Kostova, I. Manolov, G. Momekov, T. Tzanova, S. Konstantinov, and M. Karaivanova, Cytotoxic activity of new cerium (III)
complexes of bis-coumarins, Eur J Med Chem, 40 (2005), 1246‒1254.
- I. Kostova and G. Momekov, New zirconium (IV) complexes of
coumarins with cytotoxic activity, Eur J Med Chem, 41 (2006), 717‒726.
- I. Kostova, Studying plant-derived coumarins for their pharmacological and therapeutic properties as potential anticancer drugs, Expert Opin Drug Discov, 2 (2007), 1605‒1618.
- I. Kostova, I. Manolov, I. Nicolova, S. Konstantinov, and M. Karaivanova, New lanthanide complexes of 4-methyl-7-hydroxycoumarin and their pharmacological activity, Eur J Med Chem, 36 (2001), 339‒347.
- D. M. Ibrahim, J. Jumala, and F. W. Harun, Synthesis and characterization 6-hydroxy-4-metheyl-5-phenyl azo coumarins with divalent transition metal ions, IOSR J Appl Chem, 7 (2014), 27‒32.
- M. V. Kulkarni, G. M. Kulkarni, C. H. Lin, and C. M. Sun, Recent advances in coumarins and 1-azacoumarins as versatile biodynamic agents, Curr Med Chem, 13 (2006), 2795‒2818.
Copyright © 2019 Dalal M. Ibrahim et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.